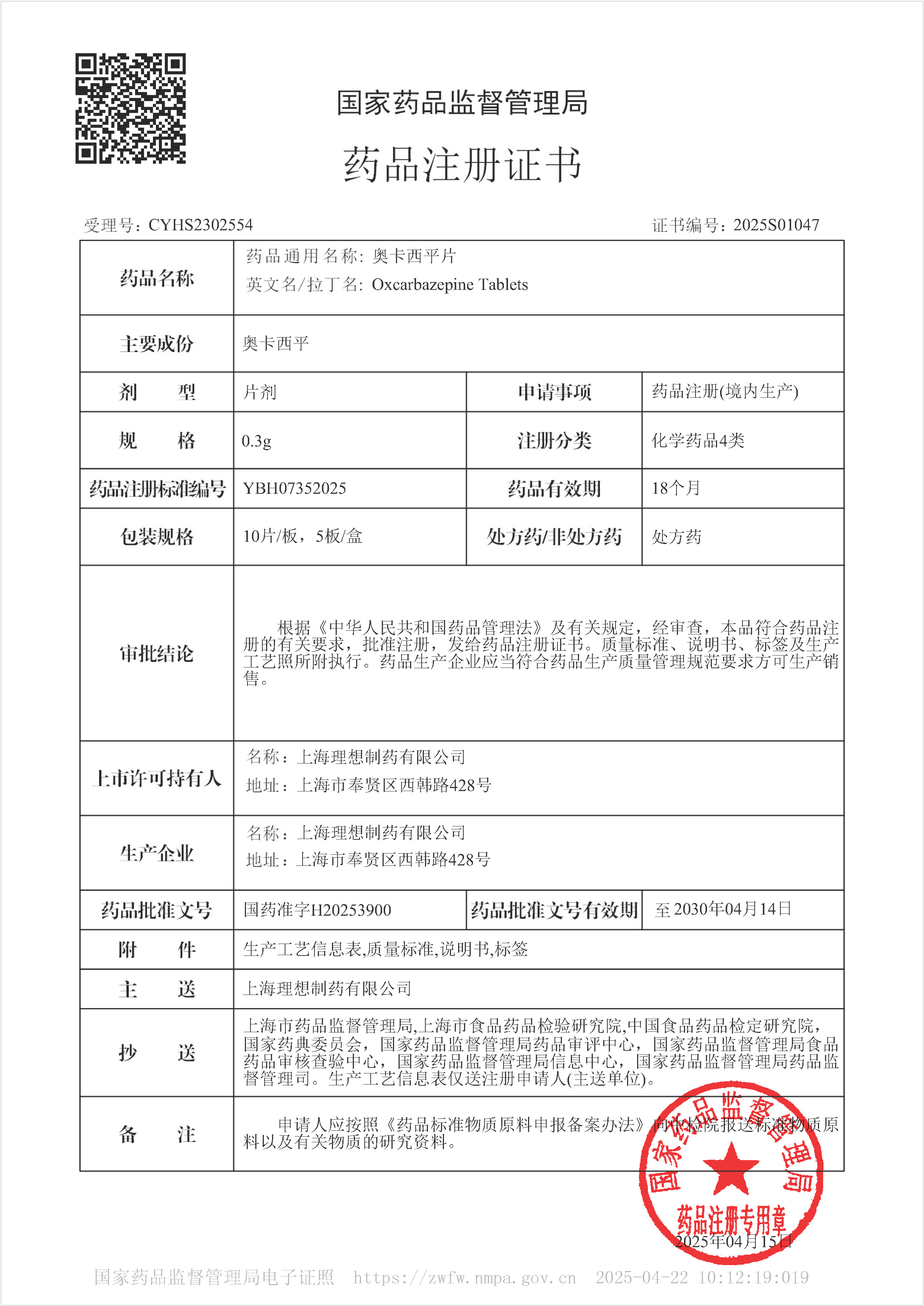
Okazepine tablets - Approved for Marketing
On April 15, 2025, our company's Okazepine tablets were approved for marketing. The product was approved according to the new registration classification of chemical drugs and deemed to have passed the consistency evaluation. This drug belongs to the national basic medicine and Class A medical insurance drugs.
Oxcarbazepine is a derivative of carbamazepine, which was approved by the US FDA as a first-line antiepileptic drug in the United States in 2000 and subsequently entered the Chinese market in 2004.
The core pharmacological effect of oxcarbazepine is exerted through the metabolite '10 monohydroxy derivative (MHD)'. The mechanism of action includes: blocking voltage sensitive sodium ion channels, stabilizing over excited neuronal cell membranes, and inhibiting abnormal discharges; Regulating calcium and potassium ion channels to enhance anticonvulsant effects; Suitable for the treatment of primary generalized tonic clonic seizures and partial seizures (with or without secondary generalized seizures), covering adult and pediatric patients aged 5 and above.