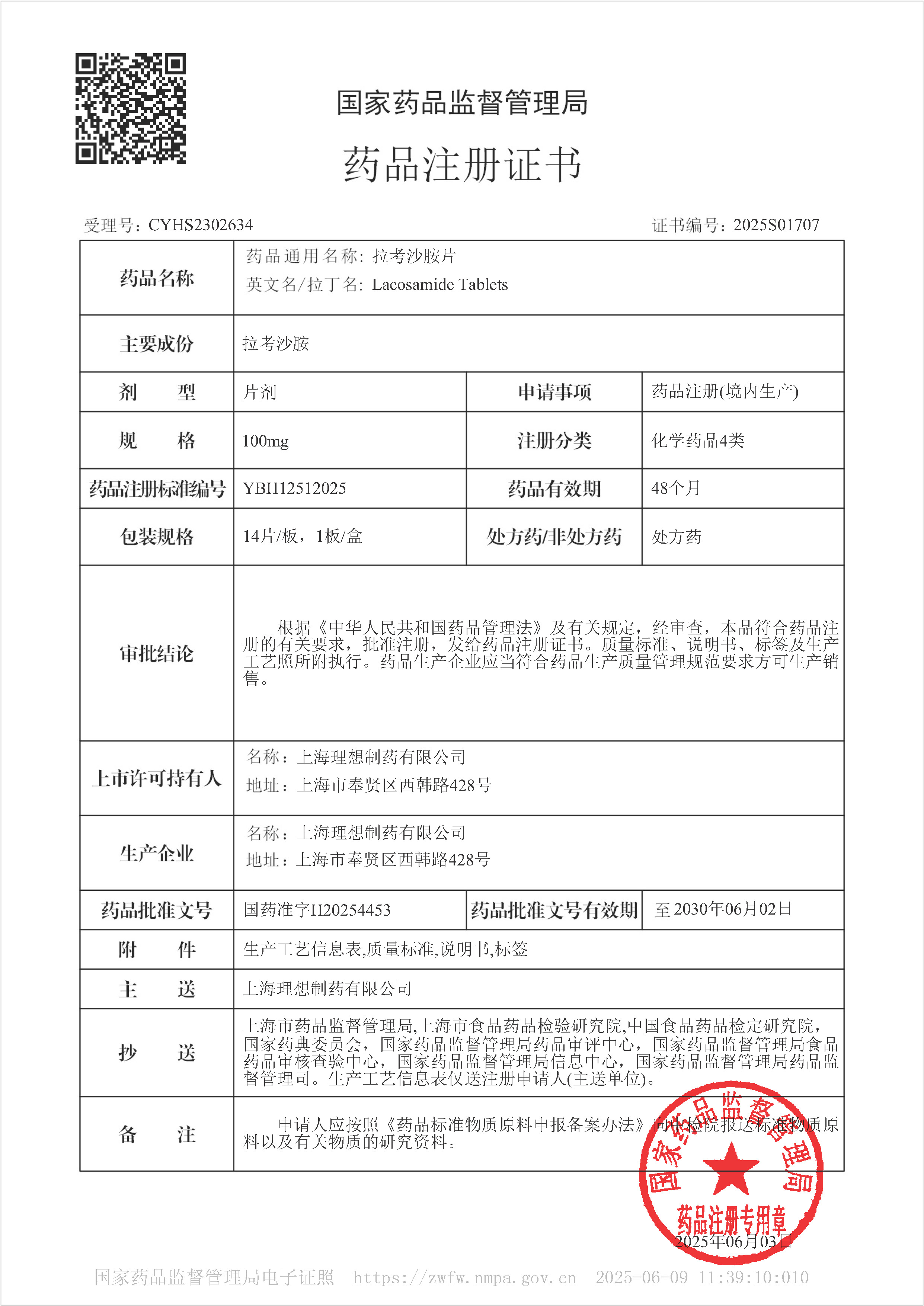
Lacosamide tablets - third-generation antiepileptic drugs
On June 3, 2025, our company's Lacosamide tablets were approved for marketing. The product was approved according to the new registration classification of chemical drugs and deemed to have passed the consistency evaluation. This medication belongs to Class B of medical insurance.
Lacosamide belongs to the third generation of antiepileptic drugs and is a novel sodium channel blocking antiepileptic drug that can selectively enhance the slow inactivation of voltage-gated sodium channels, stabilize over excited neuronal cell membranes, and inhibit repeated neuronal discharges, thereby achieving the goal of controlling epileptic seizures. This drug is generally not metabolized by the liver, has minimal impact on the liver, and generally does not react with other antiepileptic drugs, making it a commonly used medication in clinical practice. This product is suitable for monotherapy and combination therapy of partial seizures in epilepsy patients aged 4 years and above, as well as combination therapy of primary generalized tonic clonic seizures in patients aged 4 years and above.