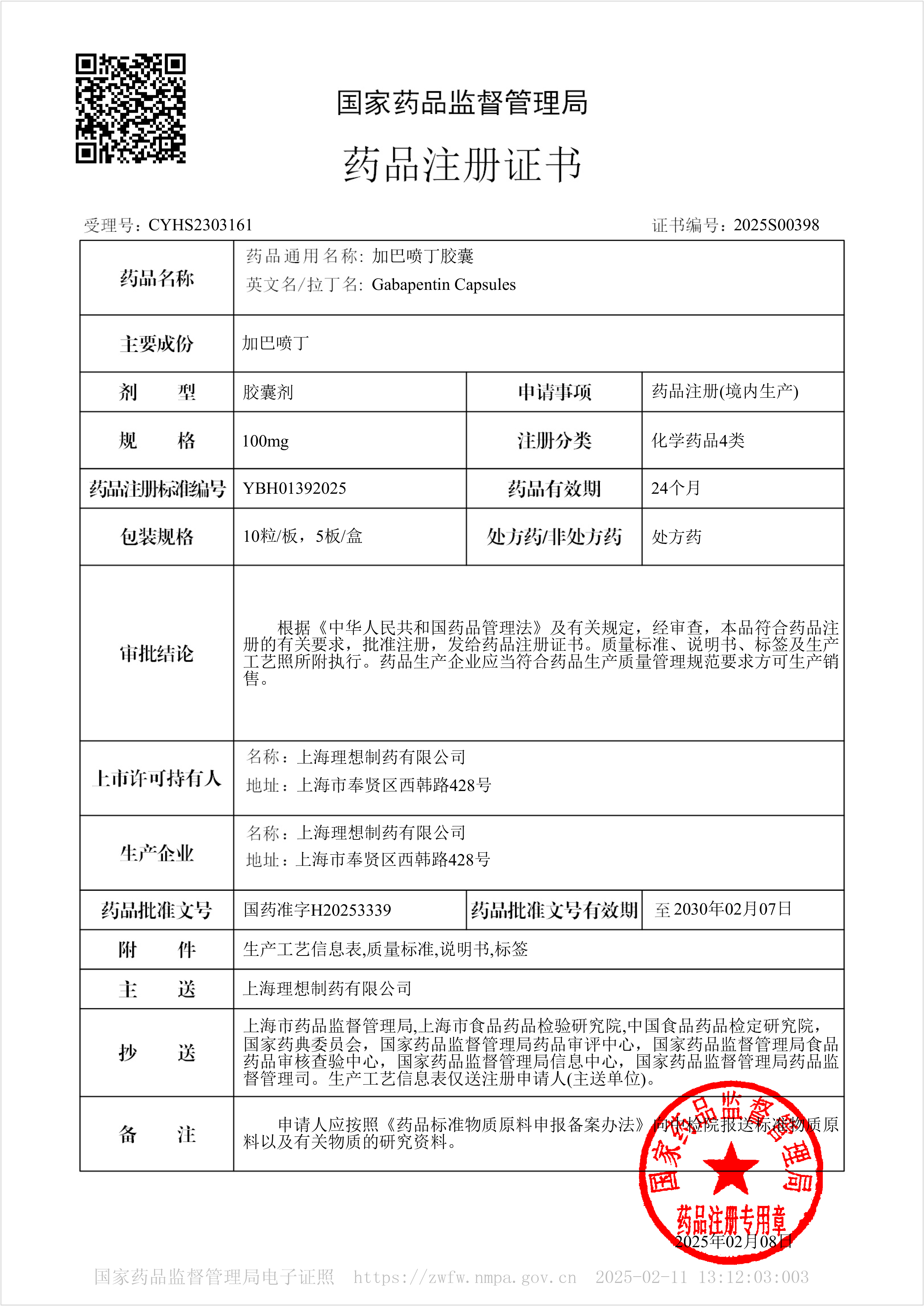
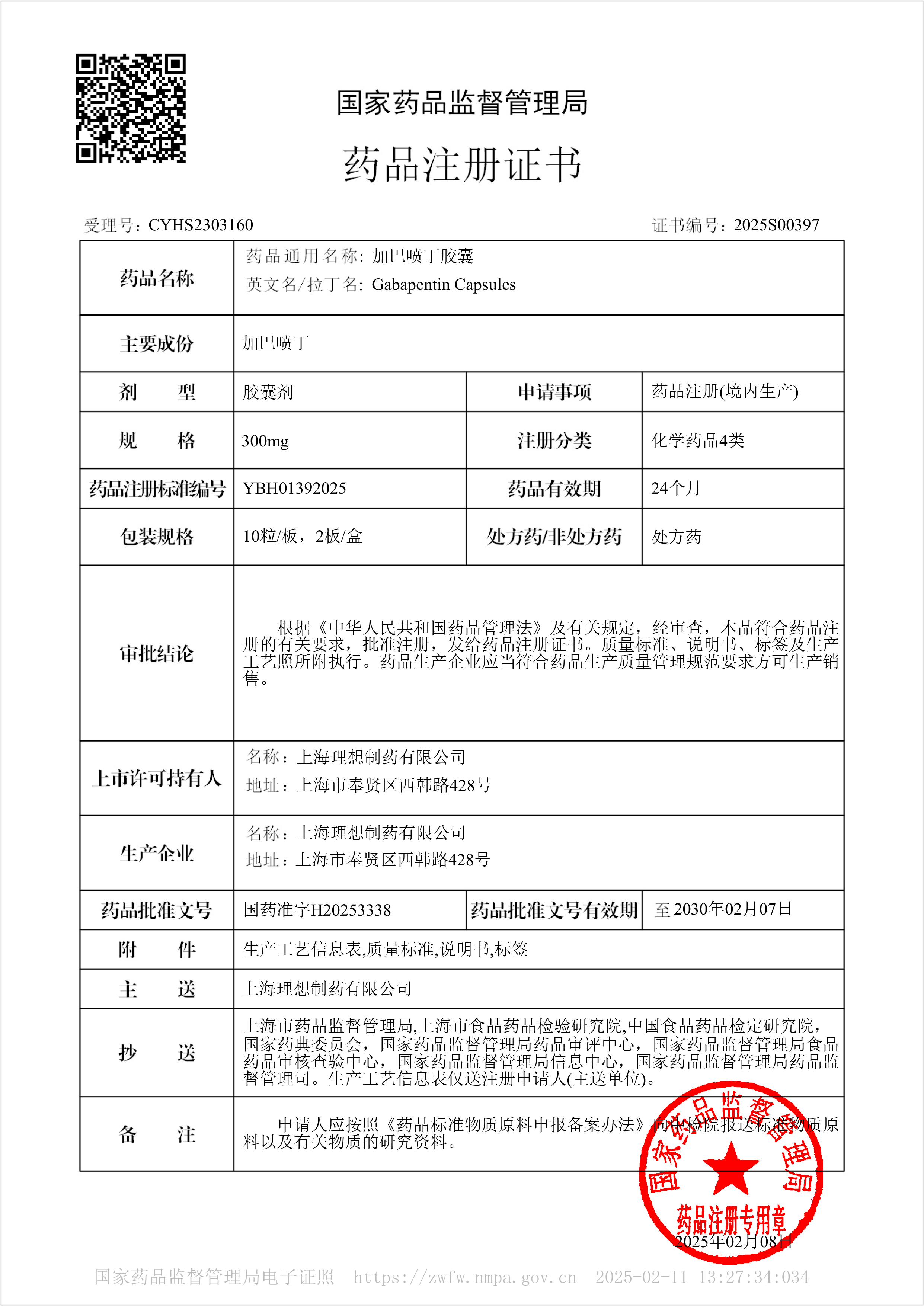
Gabapentin capsules - Approved for Marketing
On February 8, 2025, our company's another Class B antiepileptic drug, Gabapentin Capsules, was approved for market. The product was classified as a newly registered chemical drug and was deemed to have passed the consistency evaluation.
Gabapentin capsules have anti epileptic and neuropathic effects, and can be used clinically for neuropathic pain after herpes zoster infection. All pharmacological effects of gabapentin after administration are derived from the parent compound, and its metabolism in the human body is not significant, nor does it interfere with the metabolism of other commonly used antiepileptic drugs.
There is no need to monitor the blood concentration of gabapentin during the medication process. Moreover, due to the lack of significant pharmacokinetic interactions between gabapentin and other conventional antiepileptic drugs, combination therapy with this drug will not alter the plasma concentrations of these conventional antiepileptic drugs.