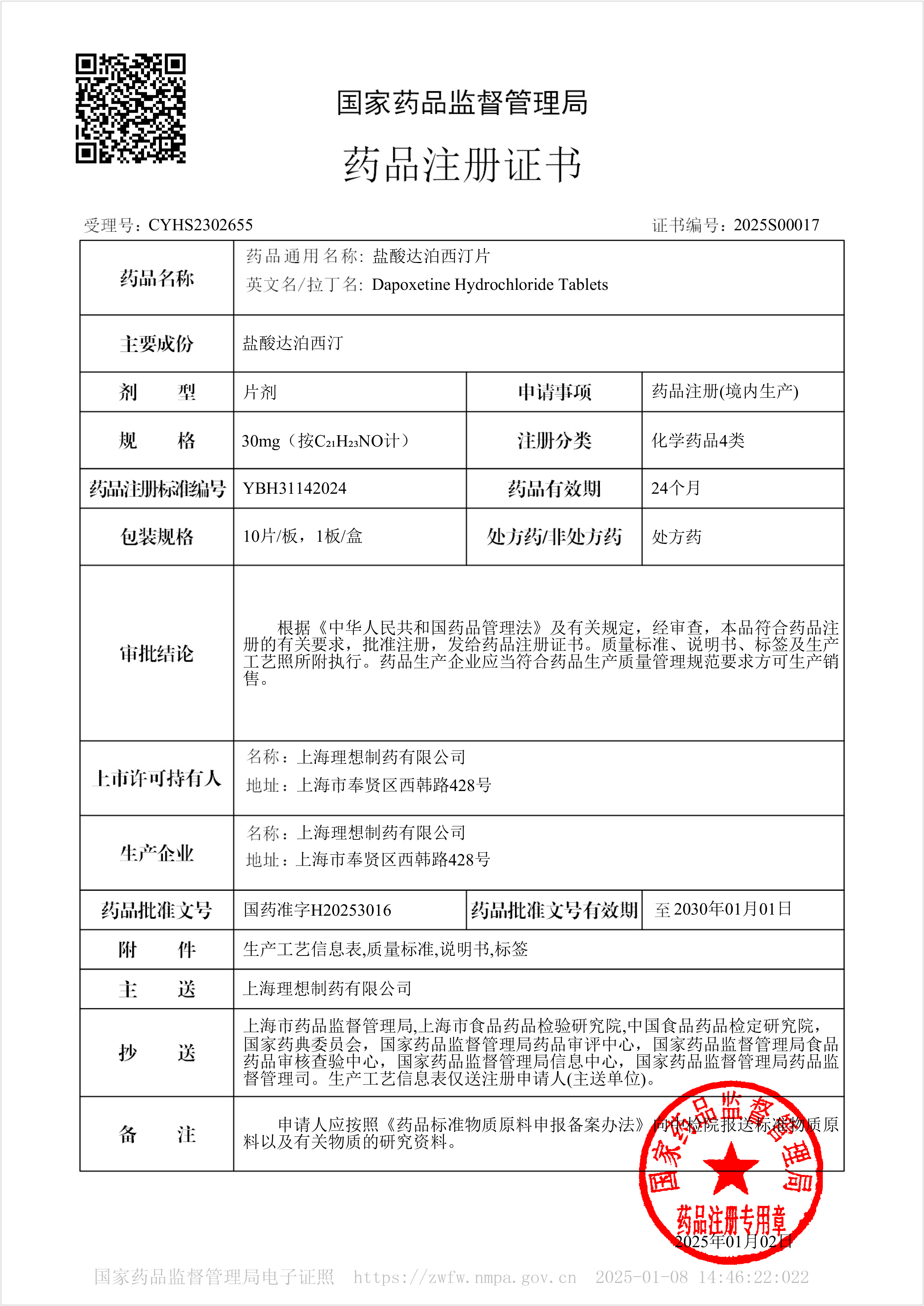
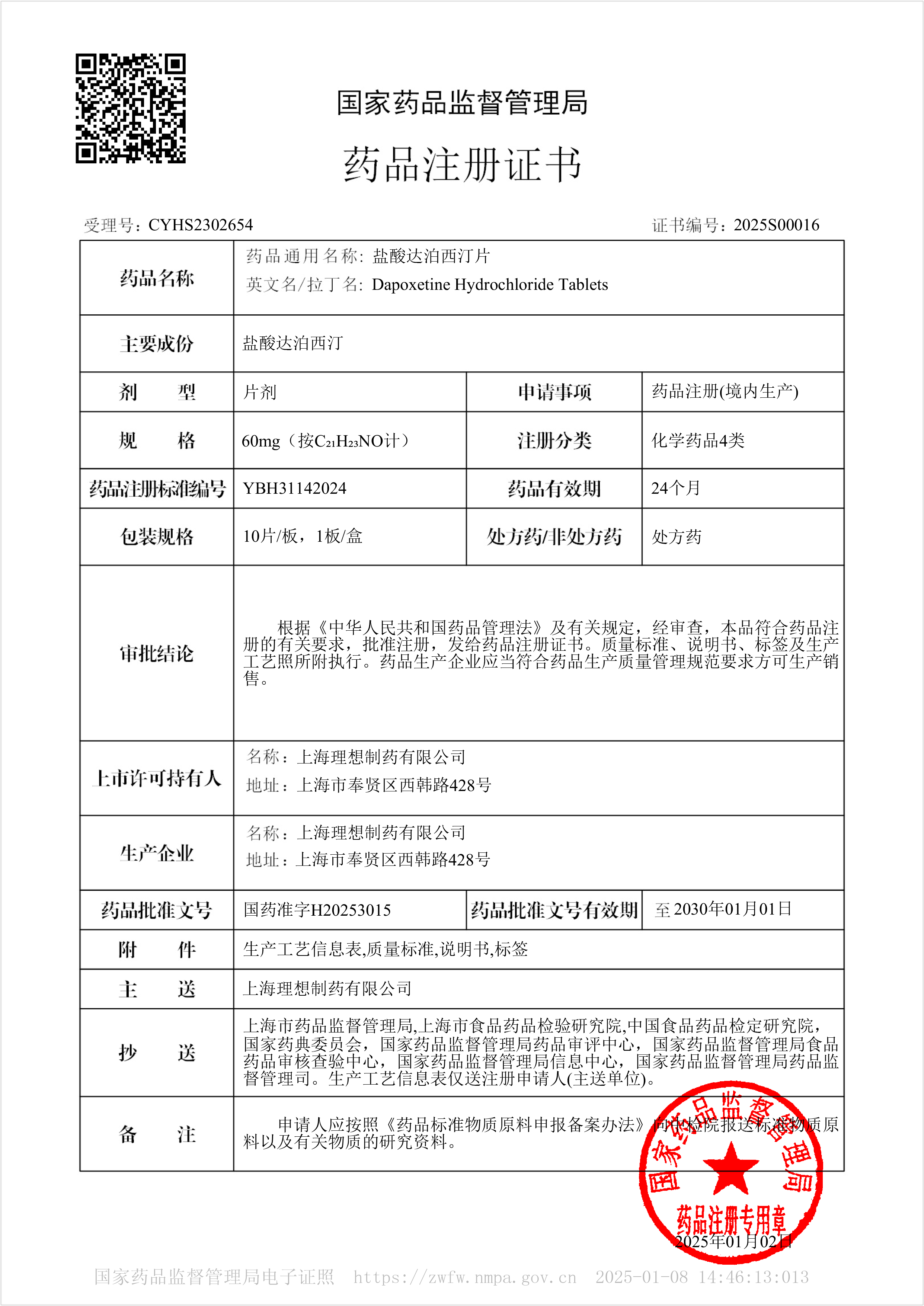
Dapoxetine Hydrochloride Tablets - Approved for Marketing
On January 2, 2025, our company's Dapoxetine Hydrochloride Tablets were approved for market. The product was approved according to the new registration classification of chemical drugs and deemed to have passed the consistency evaluation.
This product is suitable for male premature ejaculation (PE) patients aged 18 to 64 with premature ejaculation and poor ejaculation control ability.
Dapoxetine is a potent selective serotonin reuptake inhibitor (SSRI) with an IC50 of 1.12 nM. Its main metabolites are equivalent to the original drug (such as demethylated dapoxetine (IC50<1.0 nM) and dimethyl dapoxetine (IC50=2.0 nM)) or weakly effective (such as methoxy mustard dapoxetine, IC50=282 nM). Dapoxetine is unlikely to affect the pharmacokinetics of other cytochrome P450 2C19 substrates.