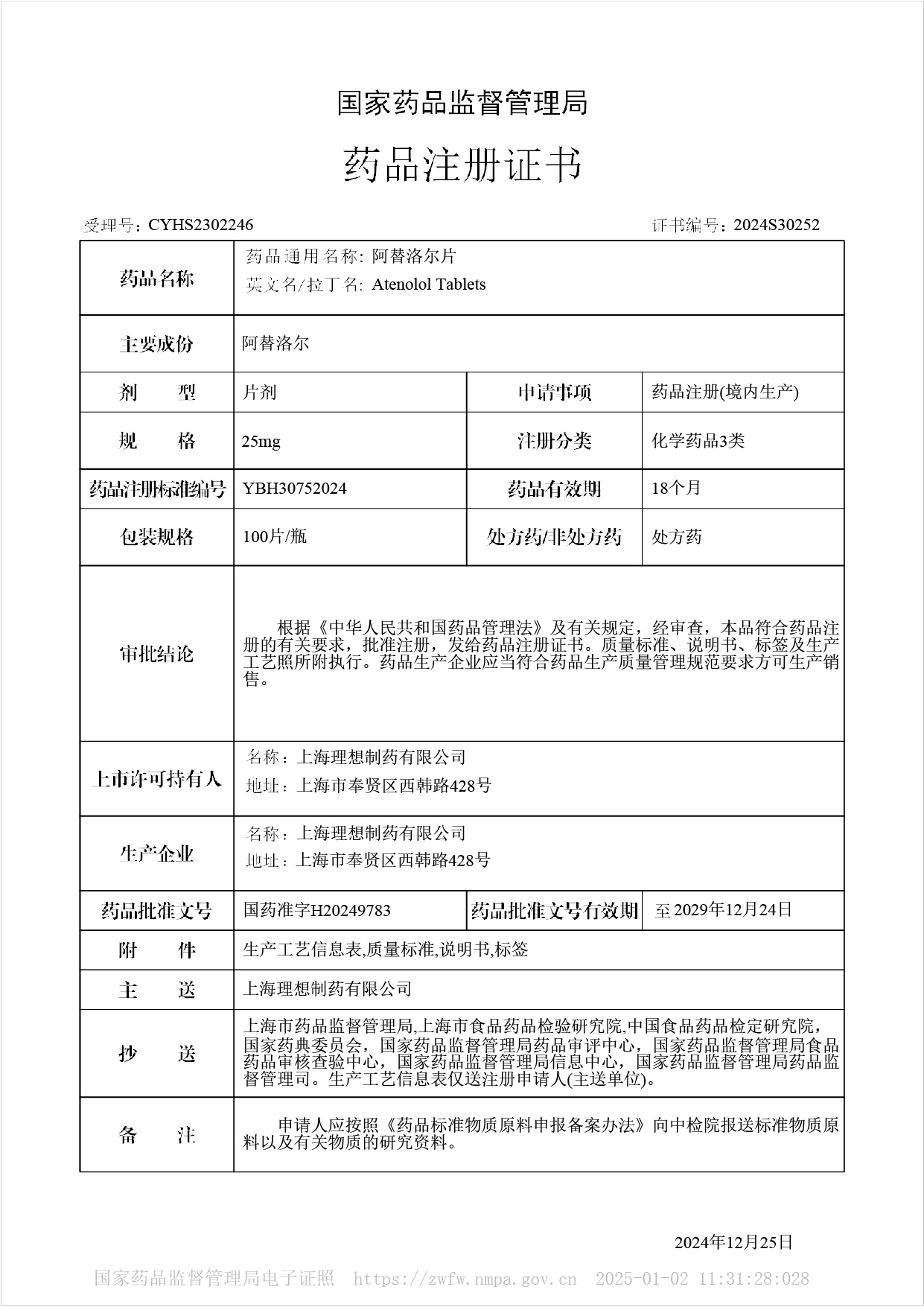
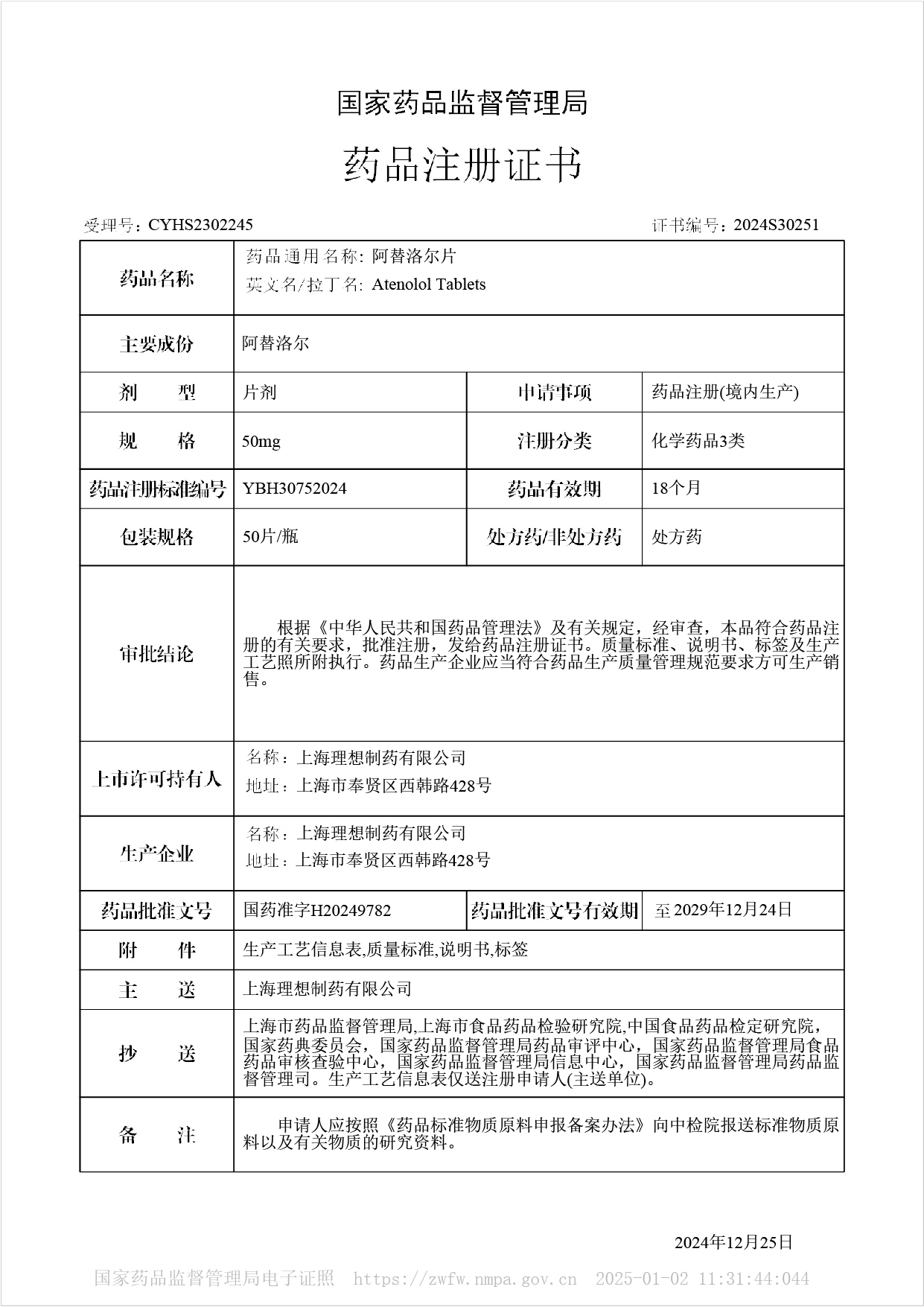
Atenolol tablets - Approved for Marketing
On December 25, 2024, our company's Atenolol tablets were approved for market, and the product was approved according to the new registration classification of chemical drugs, which is considered to have passed the consistency evaluation. This drug belongs to the national basic medicine and Class A medical insurance drugs.
Atenolol tablets, as a classic cardiovascular drug, are widely used in clinical practice. Atenolol is a selective beta adrenergic receptor blocker that does not have membrane stabilizing effects or endogenous sympathetic like activity. But it does not inhibit the bronchodilator effect of isoproterenol. It is mainly used to treat hypertension, angina pectoris, myocardial infarction, and can also be used for the treatment of arrhythmia, hyperthyroidism, and pheochromocytoma. Specific medication recommendations should be made under medical advice.